(*) and those of any age with impaired visibility
By 2030, 1 in 6 people in the world will be aged 60 years or over
World Health Organization

As the global population ages, the demand for medication will also increase. Already 10 years ago, some 87 % of people aged 75 years or more reported using prescribed medicines on a regular basis (Ageing Europe report, 2020 edition). This number is only expected to rise. As a marketer, I see this as a clear change in the target audience for pharmaceutical and life sciences companies, a change that needs to be accompanied with support on the product side for the unique requirements of older people.
This includes packaging of the products, of course, which require innovative solutions to meet the needs of a new target group while providing all crucial and mandatory information, and do so in multiple languages.
Font sizes
One of the areas where improvements may be needed is readability of the product information. While the standard clear print size for fonts to be readable is established at 12 points, this is by far not the norm in pharmaceutical leaflets, labels and others, where sizes of 7 to 9 points are more the average (this can be even smaller for food packaging, but that is a story for a different post!).
Of course, it is understandable that they are dealing with space limitations to provide a very large amount of information that is critical (and non-optional). However, due to this criticality, it is of importance that the user of the product would be able to read it properly.
Solutions like Attach-A-Leaflet®, which allows for a multi-page label to be used in a product, can help with increasing the font size while keeping packaging small.
Images/illustrations
Another aspect to help with readability, is making the leaflets easier to decipher. In general, medicine leaflets are difficult to understand for the average Joe/Jane. If the average Joe/Jane is also on the older spectrum, the difficulty increases. This is why, using images in leaflets can help translate some of the complex terms into relatable instructions to follow, making it more accesible to a wider audience.
Creating clear, easy to understand, and attractive-looking images/illustrations that support the application instructions of medicines can suppose a significant improvement for the end-user. On the packaging design sign, adding illustrations to leaflets represents an extra layer in the packaging design; however, these illustrations do not need to be changed frequently, they can be outsourced or purchased to a third party specialized in creating these sort of materials (hint: we can offer you these services, but of course, feel free to do extended research).
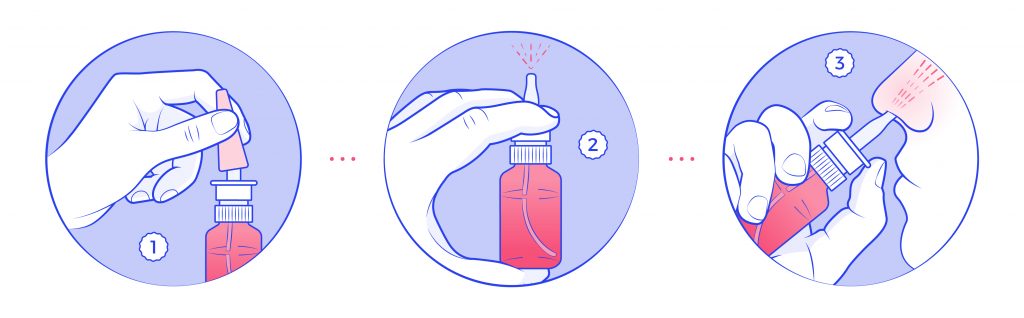
Personalized packaging
Personalization is a growing marketing trend in multiple industries, as a way to distinguish yourself from competitors. In Pharma, this personalization may come with an added value of tailoring the packaging to the individual patient needs, through the use of technology. Think of including personal medication dosages, intake instructions, or reminders to take medication.
When thinking of personalized packaging, these are some of the forms it can take:
Pre-packaged Medication
The main use of pre-packaged medication is to dispense medication into individual packages, which contain information such as the patient’s name, administration instructions and dosage in a personalized label. The main objective of these labels is to ensure patients are taking the correct (amount of) medication, so the risk of medication errors is reduced.
Electronic Medication Dispensers
In addition to pre-packaged medication, technology can also make sure that medicines are dispensed automatically to the patient at specific times, reminding patients to take medication, and providing alerts if a dose is missed. Electronic medication dispensers can be programmed to this purpose.
Smart Packaging solutions
Another idea to make packaging of medicines more accesible is to introduce the so-called Smart Packaging solutions. These innovative solution utilizes technology to provide drug users with individualized information about their medication. These can be interactive features such as audio or visual instructions such as dosage instructions and potential side effects, that are specific to the patient’s personal needs. For older people who may not be able to read small print or forget complex medication regimens, Smart packaging can give users the confidence that they are taking the correct medication at the right time, reducing the risk of medication errors.
What types of Smart packaging can be used?
- Interactive Packaging: videos, animations… in the form of augmented reality (AR) and accessed through a smartphone or tablet that can educate patients about their medication.
- Near Field Communication (NFC) Packaging: allows patients to receive information on their mobile device about dosage, potential side effects, and other important information.
- QR Code Packaging: Patients can scan the QR code, and access information about the medication, including dosage instructions and potential side effects.
Extending braille beyond the box
“The name in Braille does not have to be printed on the immediate packaging – such as blisters, ampoules and bottles, it only has to appear on the outer/secondary packaging, which is normally a carton”.
Guidance concerning the Braille requirements for labelling and the package leaflet
While not mandatory, extending braille beyond the traditional use of embossing it on the boxes, would make these more accesible to an older generation with – quite frequently – an impaired sight, giving the more independence to manage their medical needs in a safely manner . Here are some examples:
- Blisters and caps: braille added on blisters holding individual pills or medication caps would allow individuals to identify medicines by touch.
- Labels and leaflets: adding braille to the medication labels and leaflets would allow visually impaired individuals to access information such as dosage, expiry dates, warnings, contraindications, side effects…
- Vials: the use of braille in vials could be limited to information such as name, expiry date, dosage
These are some of the ideas that come to mind in order to make pharmaceutical packaging design more accesible to the needs of the ageing population (although already without thinking about a user of age, the blind and visually impaired would benefit from these improvements on a daily basis!). Ideally, everything would be implemented in the future but I would be happy to see small steps towards that goal, and legislation coming into place to make more of these “nice to haves” mandatory moving forward. What is your take on this?







